As we progress, we will present students with fun puzzles that helps sum up everything we can learn from this marvel of science. We show you how to determine the number of protons, neutrons, and electrons found in a stable atom of each element. We then look into all the compositional math that exists for these elements. These worksheets start by thoroughly explaining the information that can be found on a single atom of an element on the table. While the atomic radius measures the size of a neutral atom, the ionic radius gauges the size of an electrically charged atom. The worksheets range from finding basic information about particular elements that are found in nature to working on determine atomic operations such as neutron numbers. Atom sizes are relative to the largest atom, cesium. 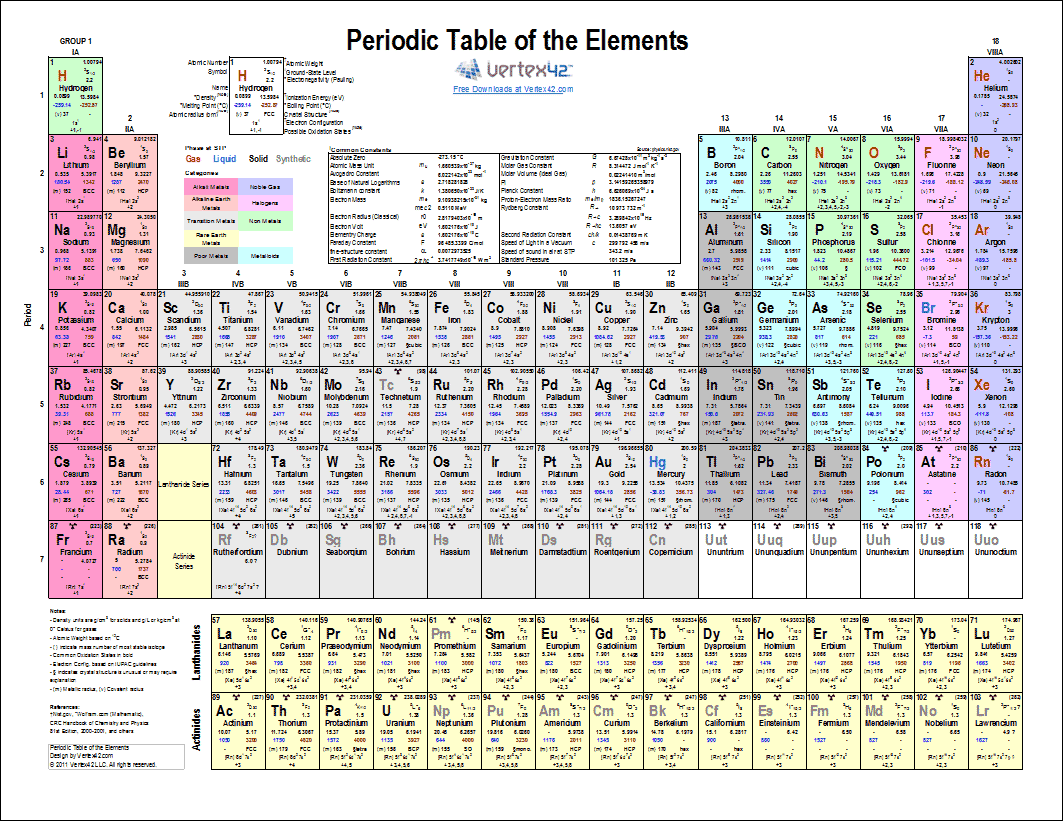
This really helps us also write the chemical formulas for compounds and balance chemical equations. As you progress from right to left in a row of elements several trends emerge: the amount of energy required to remove an electron increases, the radius of an atom of the element decreases, as well as the increased tendency to attract a pair electrons. Elements that exist in the same column of the table tend to not only similar chemical properties, but also form similarly charged ions. Students can use the Periodic Table to help them predict the outcome of chemical reactions and the stability of compounds in the modern science classroom. The elements on the table can be found at room temperature in all of the various phases. Ptables new, up-to-date periodic table PDF and wide periodic table PDF are layered so you can choose exactly what you want to print, and are the perfect companion to the periodic table classroom poster. Elements on the table are ordered by their number of protons which is equivalent to their atomic number. He also used it to predict the properties of elements that were either not discovered yet or not normally found in nature. He showed that a certain arrangement of elements could lead us to understand the properties of other elements that were related to each other. Therefore, there are various non-equivalent definitions of atomic radius.Noted scientist Dmitri Mendeleev was touted as creating the first version of the Periodic Table. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Gold atom is 136pm (covalent radius). 
Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. This periodic table contains the oxidation numbers of the elements as well as element numbers, symbols, names, and atomic weights. 
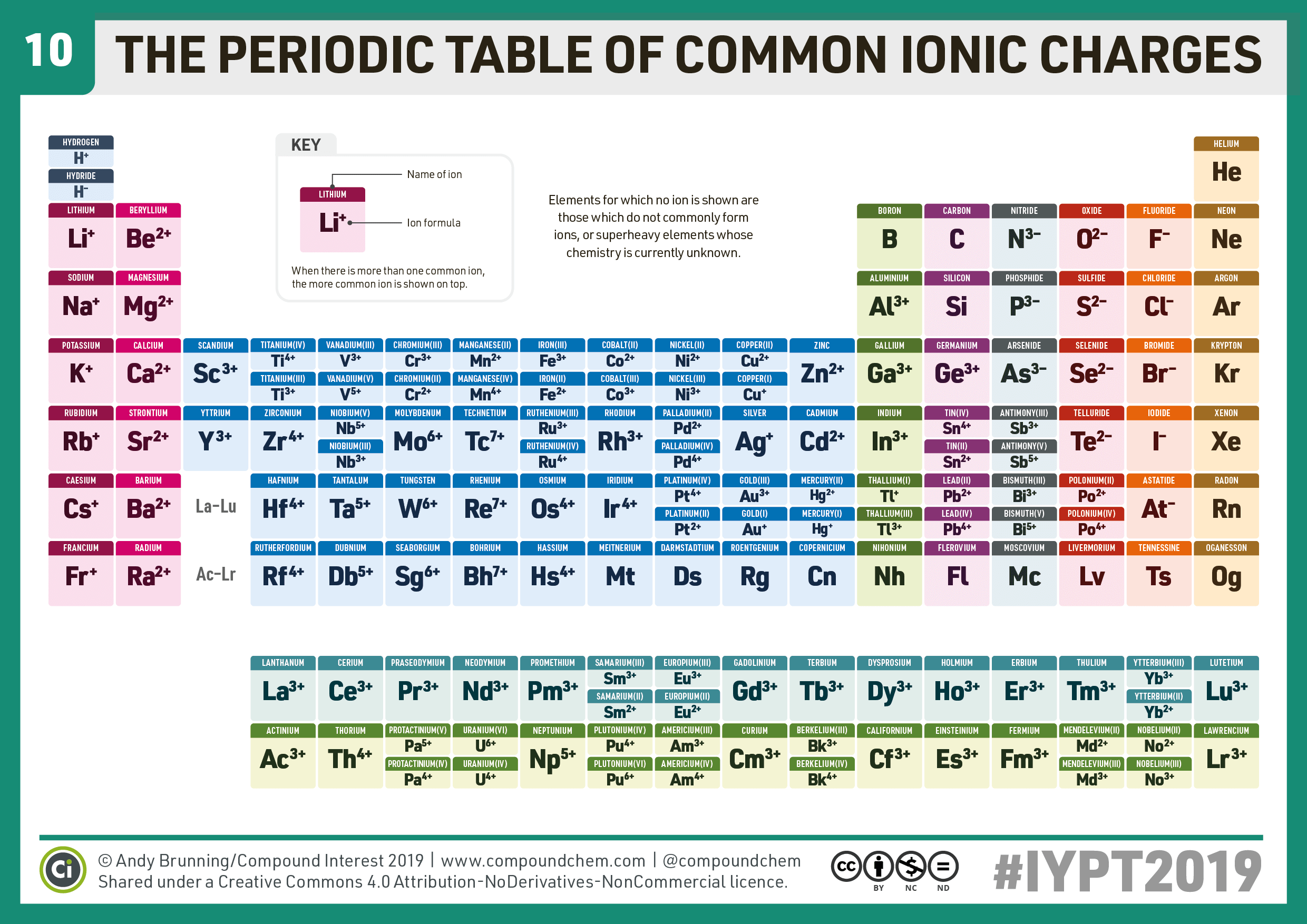
Halogens, on the other hand, have an oxidation state of -1. For example, iron common has an oxidation number of +2 or +3. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. However, most metals are capable of multiple oxidation states. Mass numbers of typical isotopes of Gold are 197. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. For example, a sodium atom usually has a +1 charge, while oxygen most often has a -2 charge, and chlorine usually has a -1 charge. The valence of an atom is its oxidation state. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. This printable periodic table contains the atomic number, element symbol, element name, atomic weights and most common valence charges. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Gold is a chemical element with atomic number 79 which means there are 79 protons in its nucleus. Common Polyatomic ions Ammonium +1NH 4 Hydroxide OH-1 Nitrate -NO 3 1 Sulfate - SO 4 2 Nitrite NO-1-Sulfite SO 2 ClO 3-1 CO 3-2 Cyanide CN-1 Phosphate PO 4 3 n sulfate HSO 4-1 Phosphite PO-3 Hydrogen carbonate HCO-1 - Peroxide O 2 (bicarbonate) HPO 4-2 Periodic Table & Polyatomic Ions Chart. Atomic Number – Protons, Electrons and Neutrons in Gold
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
